Cochlear Implants
Cochlear Implants
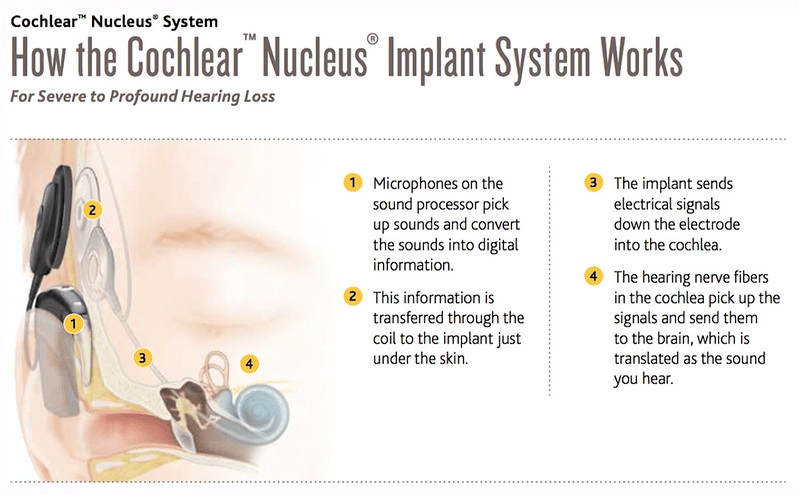
Cochlear implants are U.S. Food and Drug Administration (FDA)-approved, surgically-implanted medical devices that treat hearing loss. Cochlear implants replace the function of a damaged inner ear (cochlea) and are designed to mimic natural hearing.
Who is a candidate?
An audiologist will determine your candidacy based on your audiometric findings. A diagnostic hearing test will be performed as well as specialized speech testing. Here are some of the guidelines:
- Individuals 12 months of age or older
- Moderate to profound sensorineural hearing loss in both ears
- Limited benefit from amplification defined by preoperative test scores of ≤ 50% sentence recognition in the ear to be implanted and ≤60% in the opposite ear or binaurally
To learn more, please visit the Cochlear website at www.cochlear.com/us


